Effectiveness of Ritonavir-Boosted Protease Inhibitor Monotherapy in Clinical Practice Even with Previous Virological Failures to Protease Inhibitor-Based Regimens | PLOS ONE

Ritonavir boosting of low-dose danoprevir (RG7227; ITMN-191), HCV NS3/4A protease inhibitor, results in robust reduction in HCV RNA at lower exposures than provided by unboosted regimens

Structures of the boosters (CYP inhibitors) ritonavir and cobicistat,... | Download Scientific Diagram

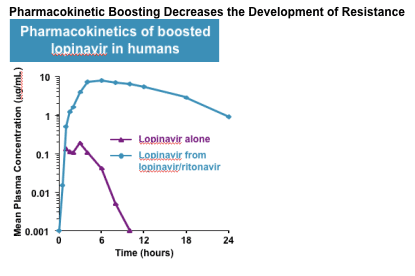
Lopinavir–ritonavir super-boosting in young HIV-infected children on rifampicin-based tuberculosis therapy compared with lopinavir–ritonavir without rifampicin: a pharmacokinetic modelling and clinical study - The Lancet HIV

Adrenal insufficiency due to ritonavir-triamcinolone drug–drug interaction without preceding Cushing's syndrome - S Noe, H Jaeger, S Heldwein, 2018

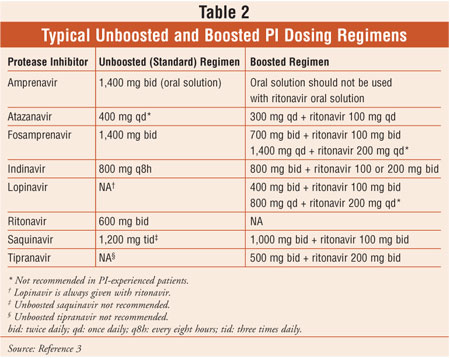
Overview of boosted protease inhibitors in treatment-experienced HIV-infected patients. | Semantic Scholar

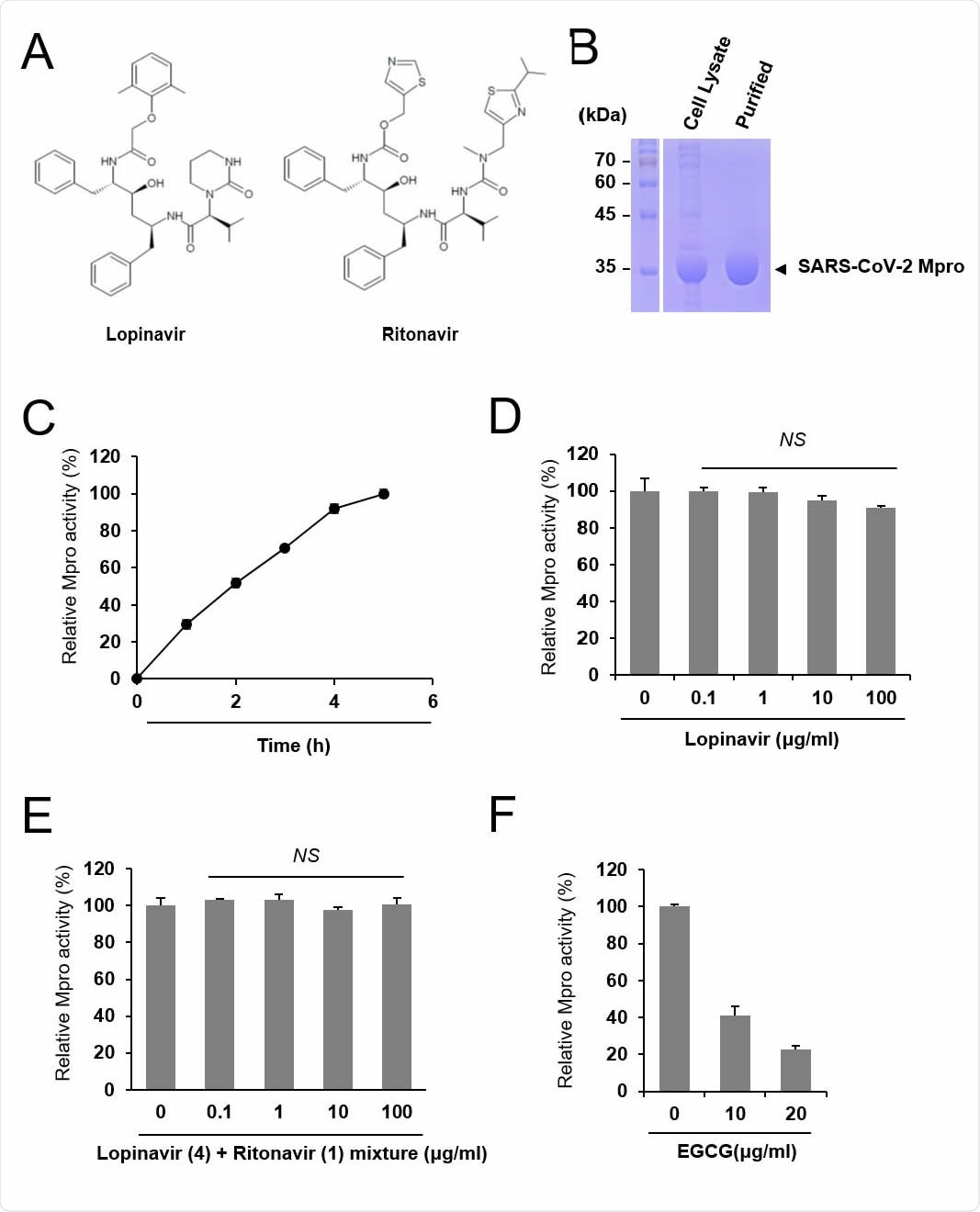
High rate of major drug–drug interactions of lopinavir–ritonavir for COVID-19 treatment | Scientific Reports

Biomedicines | Free Full-Text | Management of Antiretroviral Therapy with Boosted Protease Inhibitors—Darunavir/Ritonavir or Darunavir/Cobicistat

Dual therapy with ritonavir-boosted protease inhibitor (PI) plus lamivudine versus triple therapy with ritonavir-boosted PI plus two nucleos(t)ide reverse-transcriptase inhibitor in HIV-infected patients with viral suppression - ScienceDirect

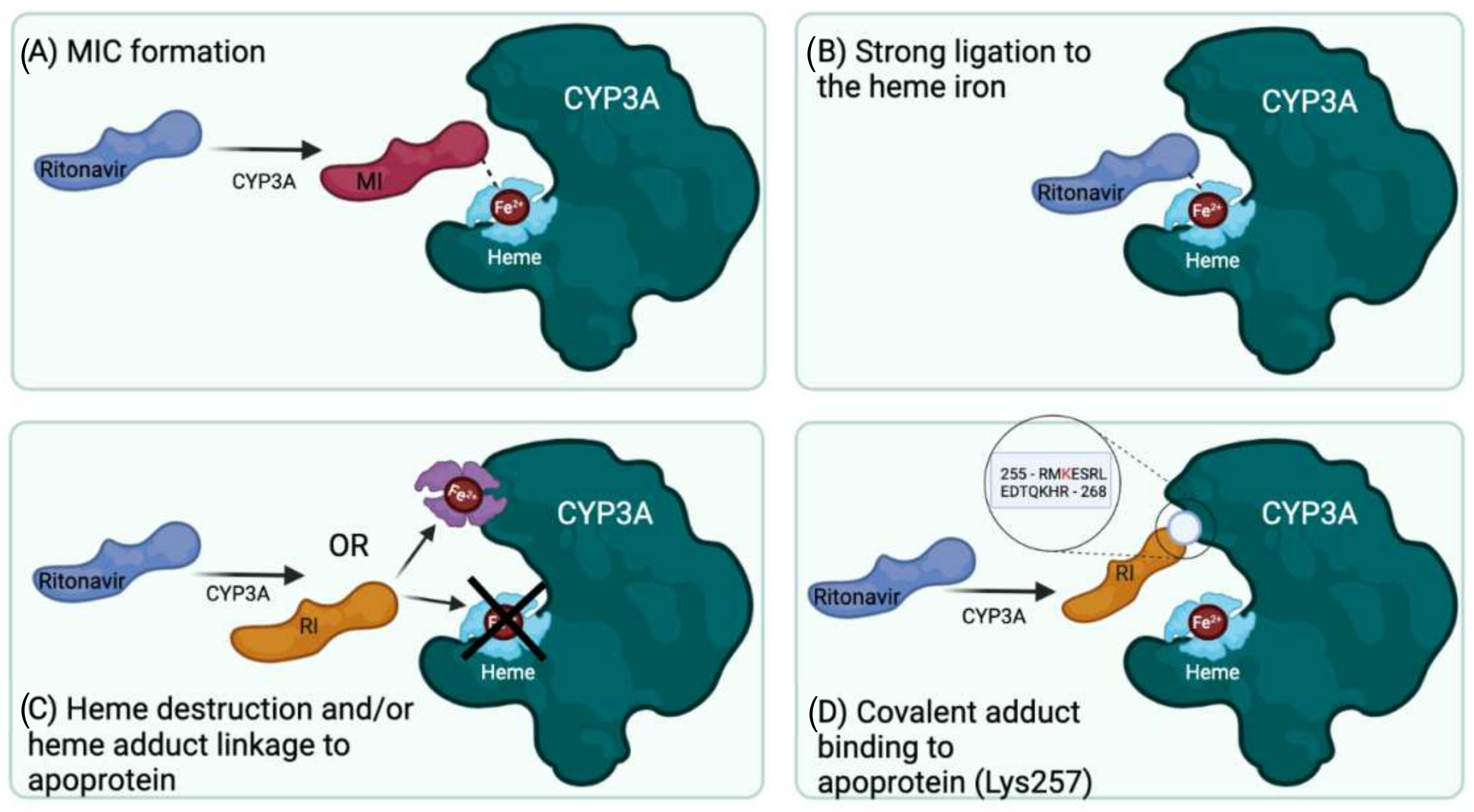
The inhibitory and inducing effects of ritonavir on hepatic and intestinal CYP3A and other drug-handling proteins - ScienceDirect