Lantus (Insulin Glargine Injection) 100 units/ml Solostar - 3 ml Prefilled Pen | Pharmacy Diabetes & Insulin | PetSmart
Mylan Pharmaceuticals Inc., a Viatris Company, Issues Voluntary Nationwide Recall of One Batch of Insulin Glargine (Insulin glargine-yfgn) Injection Pens, 100 units/mL (U-100), Due to the Potential of Missing Labels on Some

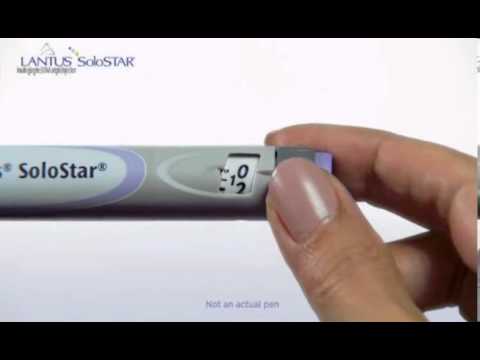
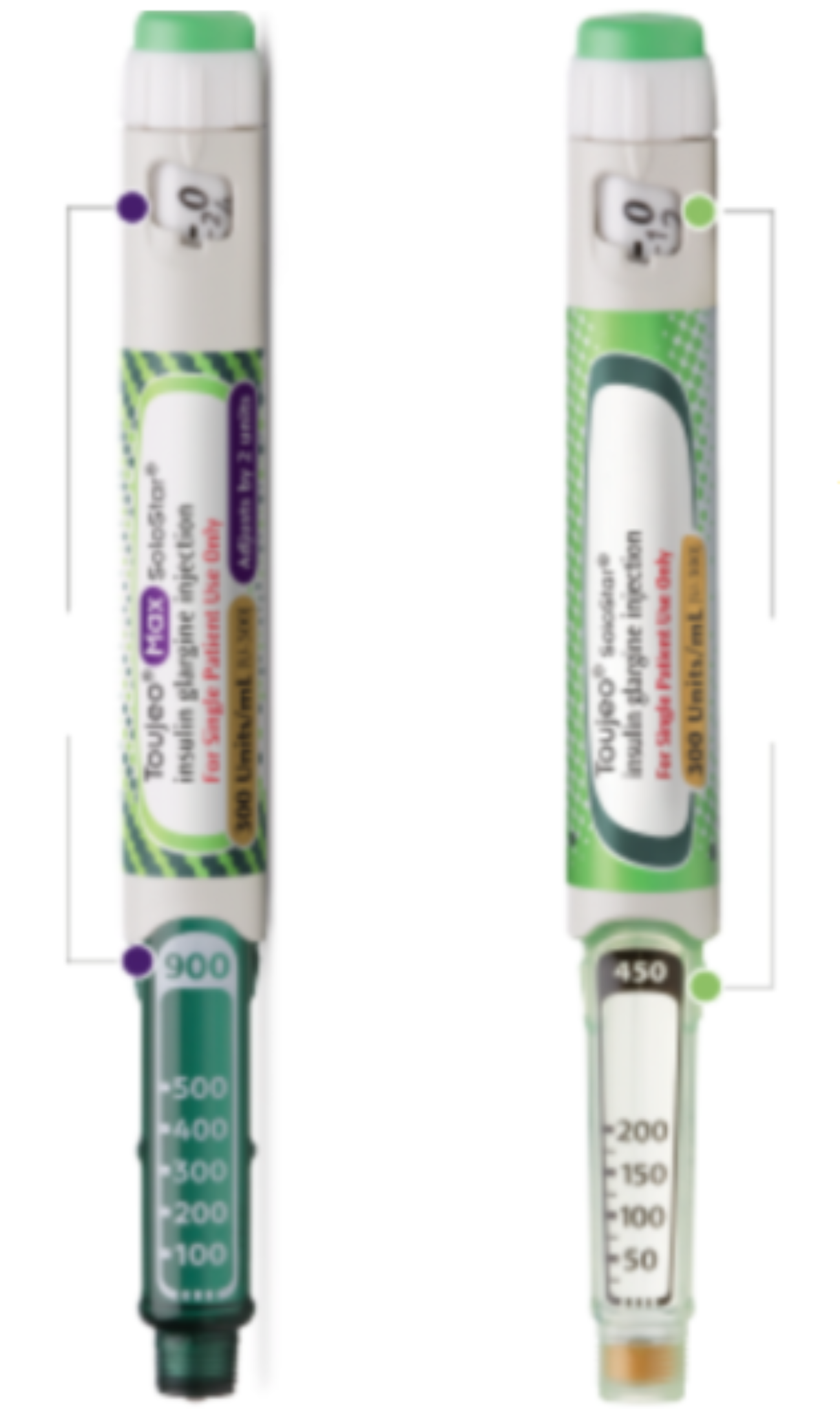
Dose Disparity: The Improper Use of an Insulin Glargine Pen: A Case Report - Natalie Nienhaus, Jennifer Alisha Rivas, Kimberly A. B. Cauthon, 2021
Lantus Solostar U-100 Insulin subcutaneous: Uses, Side Effects, Interactions, Pictures, Warnings & Dosing - WebMD