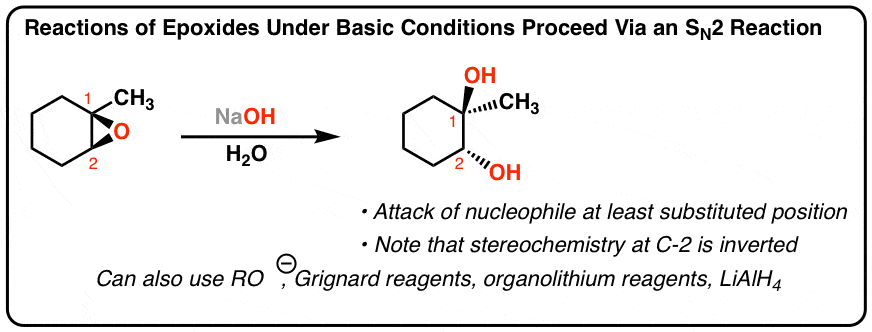
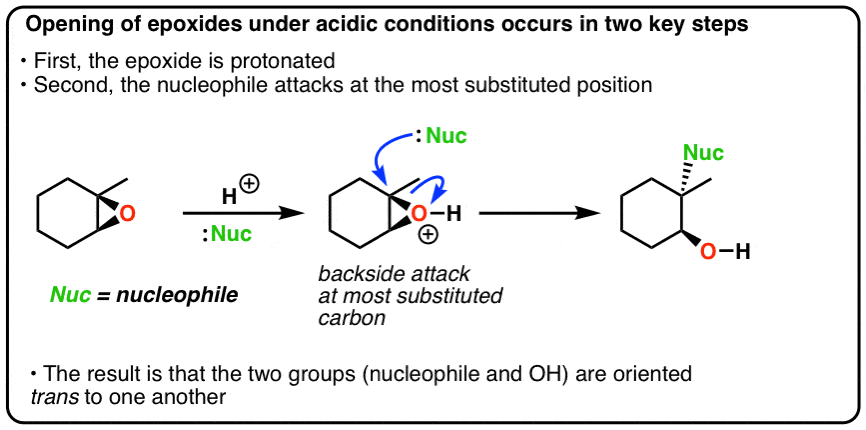
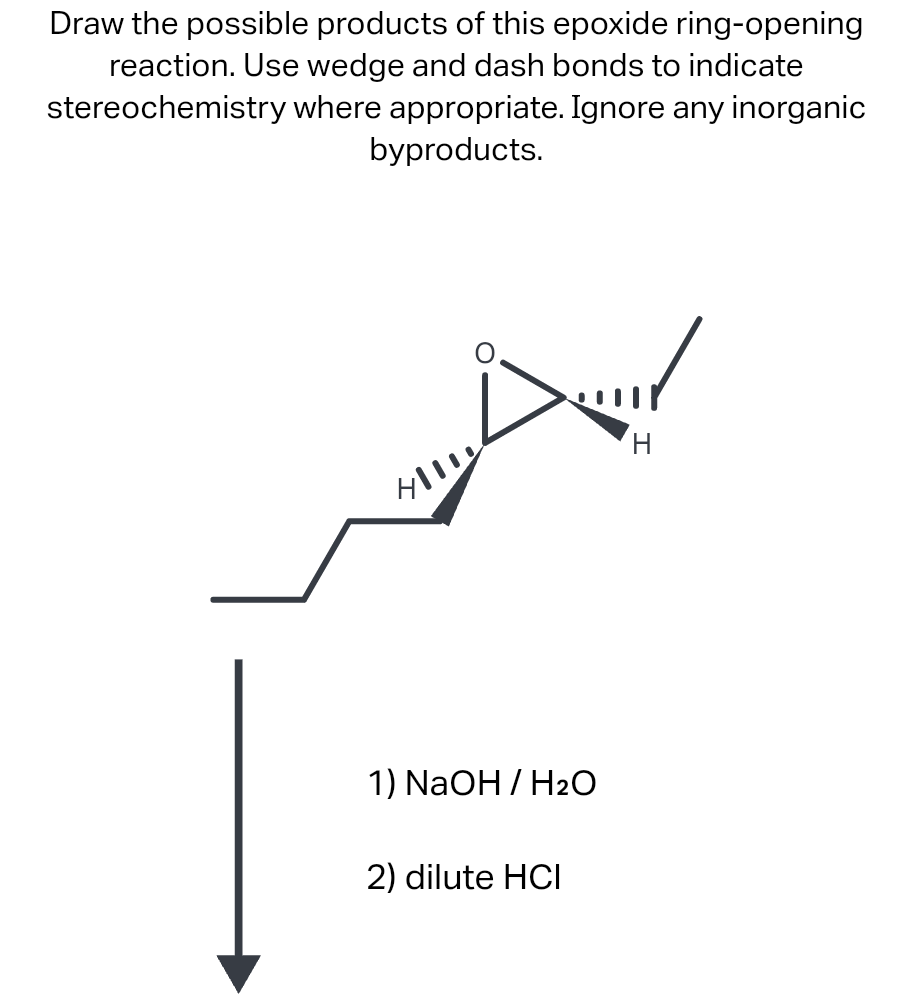
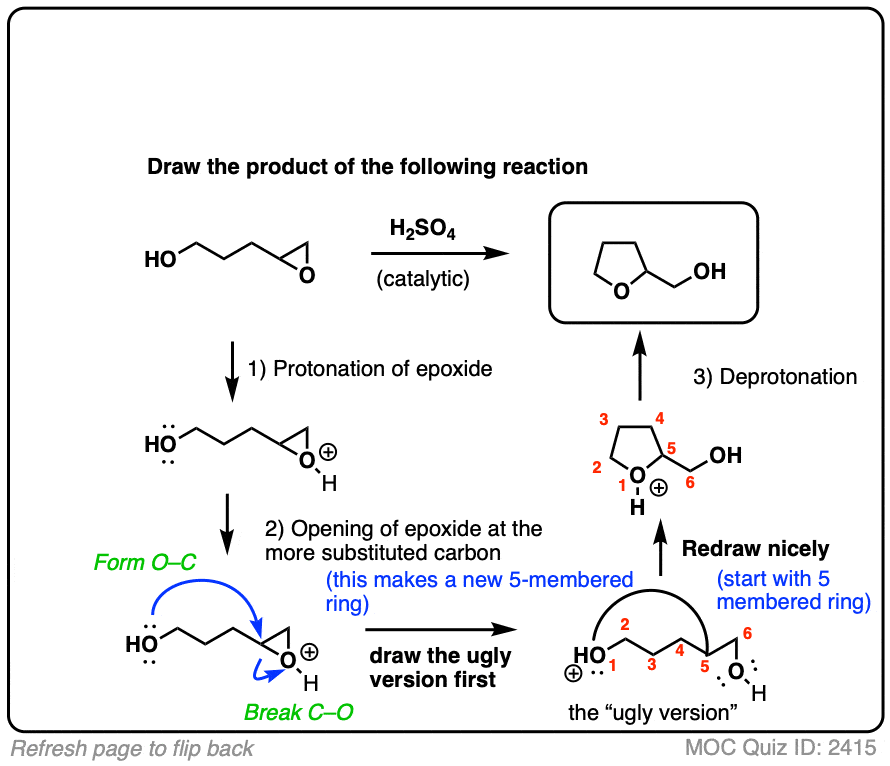
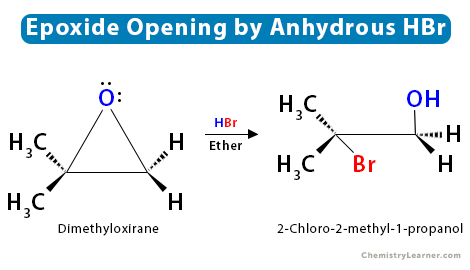
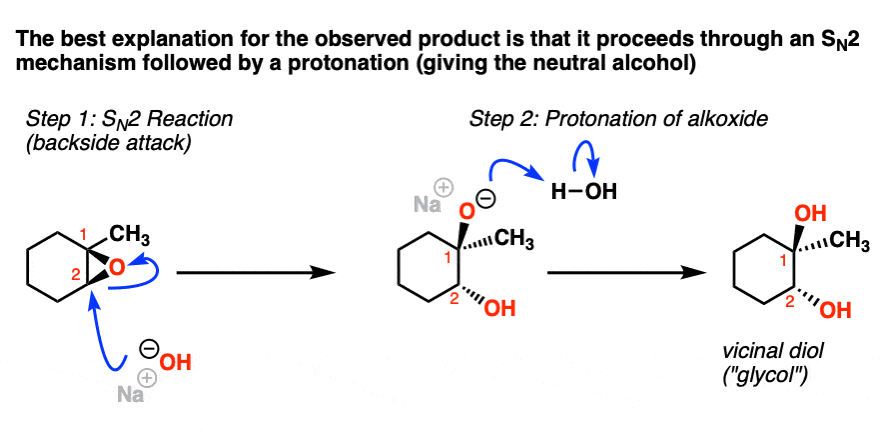
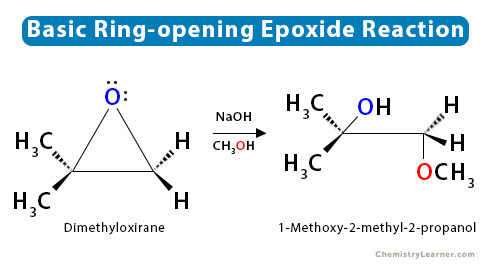
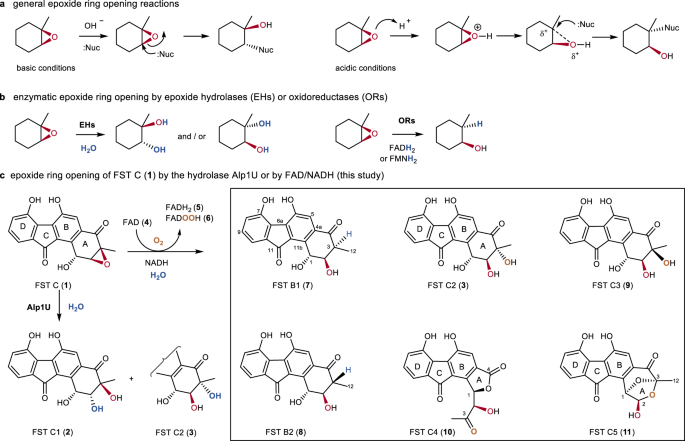
Regioselectivity of Epoxide Ring‐Openings via SN2 Reactions Under Basic and Acidic Conditions - Hansen - 2020 - European Journal of Organic Chemistry - Wiley Online Library
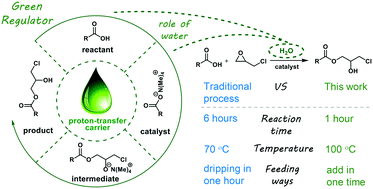
Remarkable improvement of epoxide ring-opening reaction efficiency and selectivity with water as a green regulator - Reaction Chemistry & Engineering (RSC Publishing)

Experimental and Computational Studies of Microwave-Assisted, Facile Ring Opening of Epoxide with Less Reactive Aromatic Amines in Nitromethane | ACS Omega
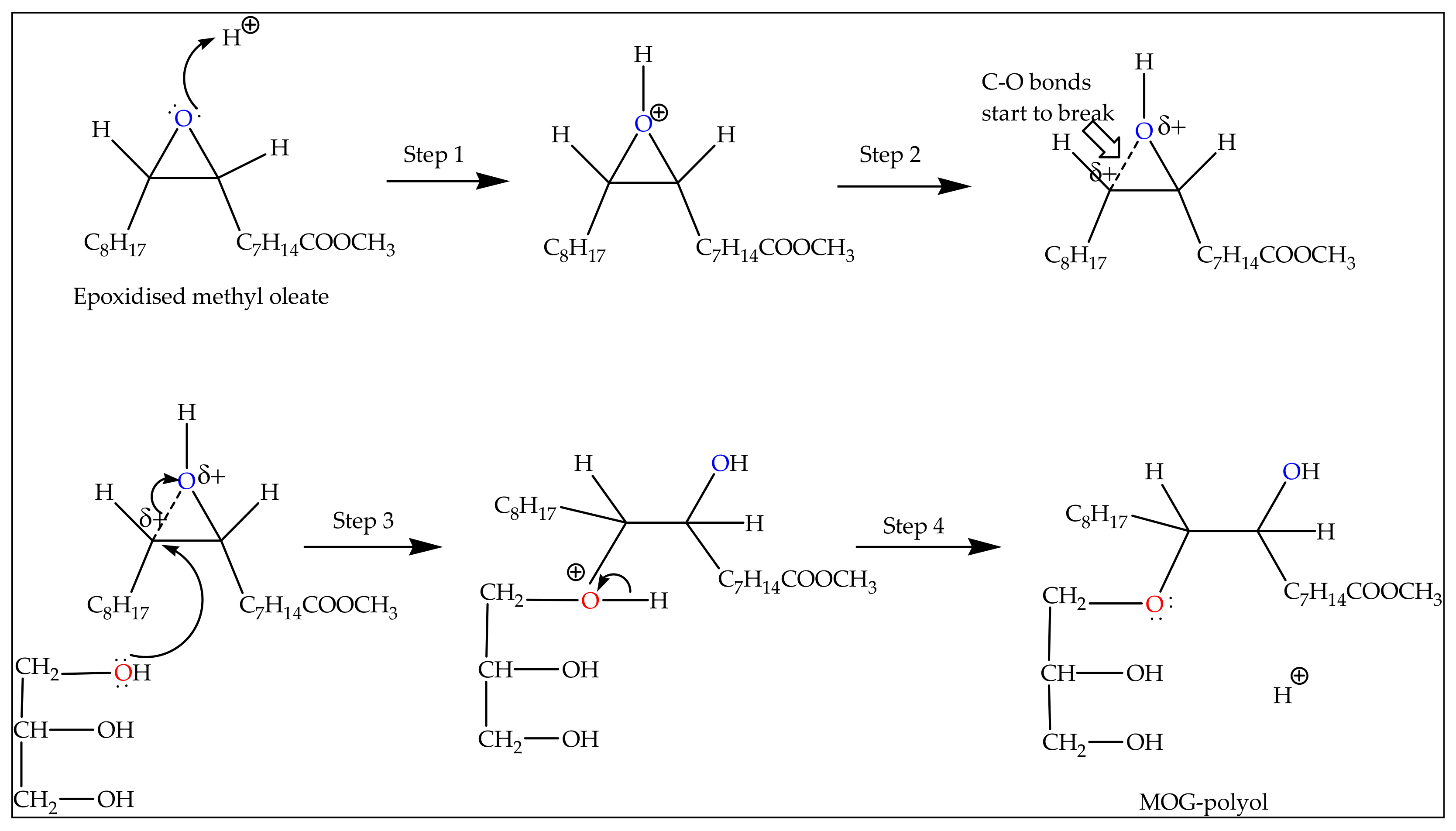
Molecules | Free Full-Text | Optimisation of Epoxide Ring-Opening Reaction for the Synthesis of Bio-Polyol from Palm Oil Derivative Using Response Surface Methodology

Regiodivergent Hydroborative Ring Opening of Epoxides via Selective C–O Bond Activation | Journal of the American Chemical Society

Brønsted Acid‐Catalysed Epoxide Ring‐Opening Using Amine Nucleophiles: A Facile Access to β‐Amino Alcohols - Tyagi - 2022 - Chemistry – An Asian Journal - Wiley Online Library

![Epoxide ring opening using methanol [70]. | Download Scientific Diagram Epoxide ring opening using methanol [70]. | Download Scientific Diagram](https://www.researchgate.net/publication/337674784/figure/fig5/AS:905644148289537@1592933799869/Epoxide-ring-opening-using-methanol-70.jpg)