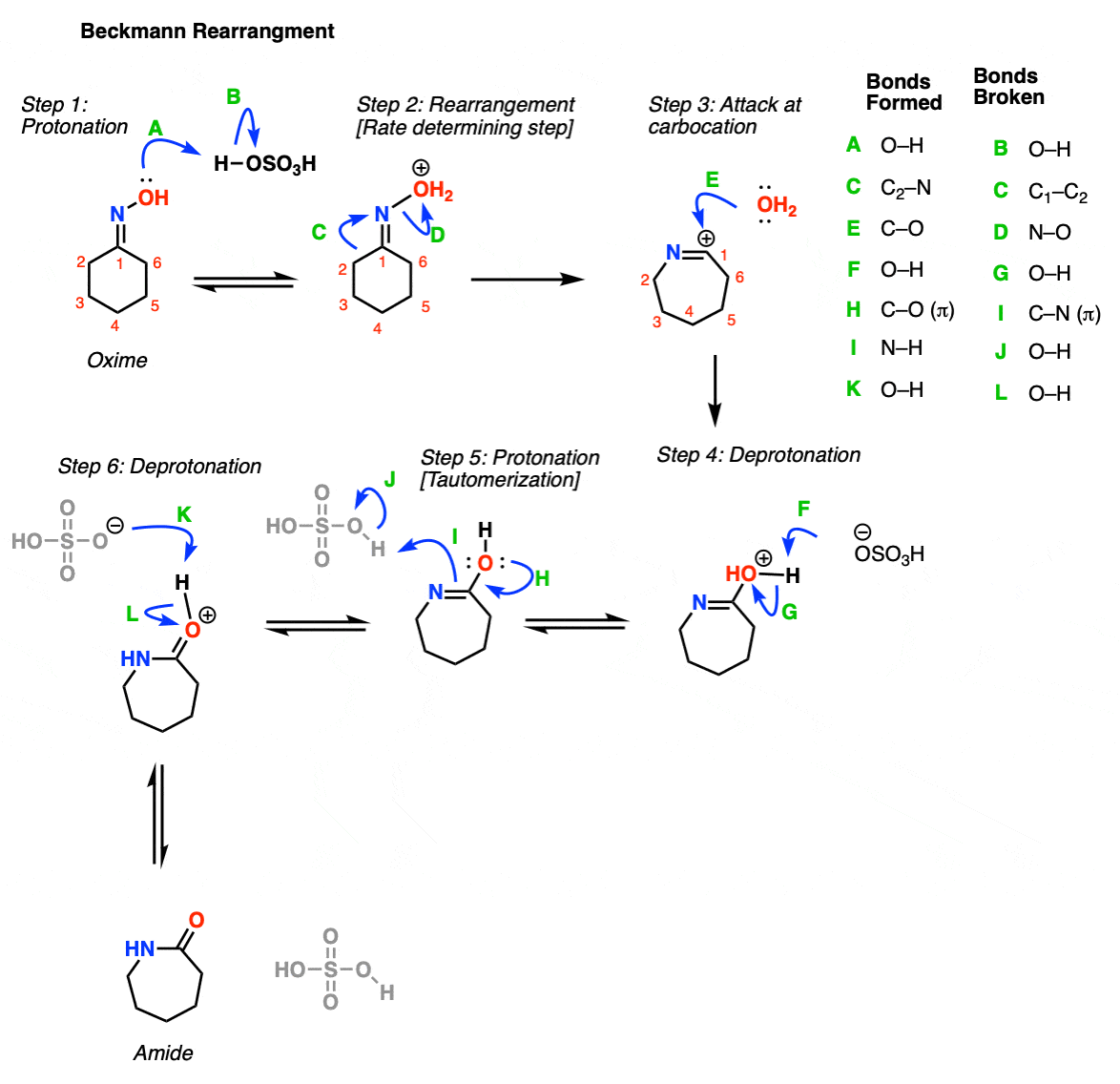
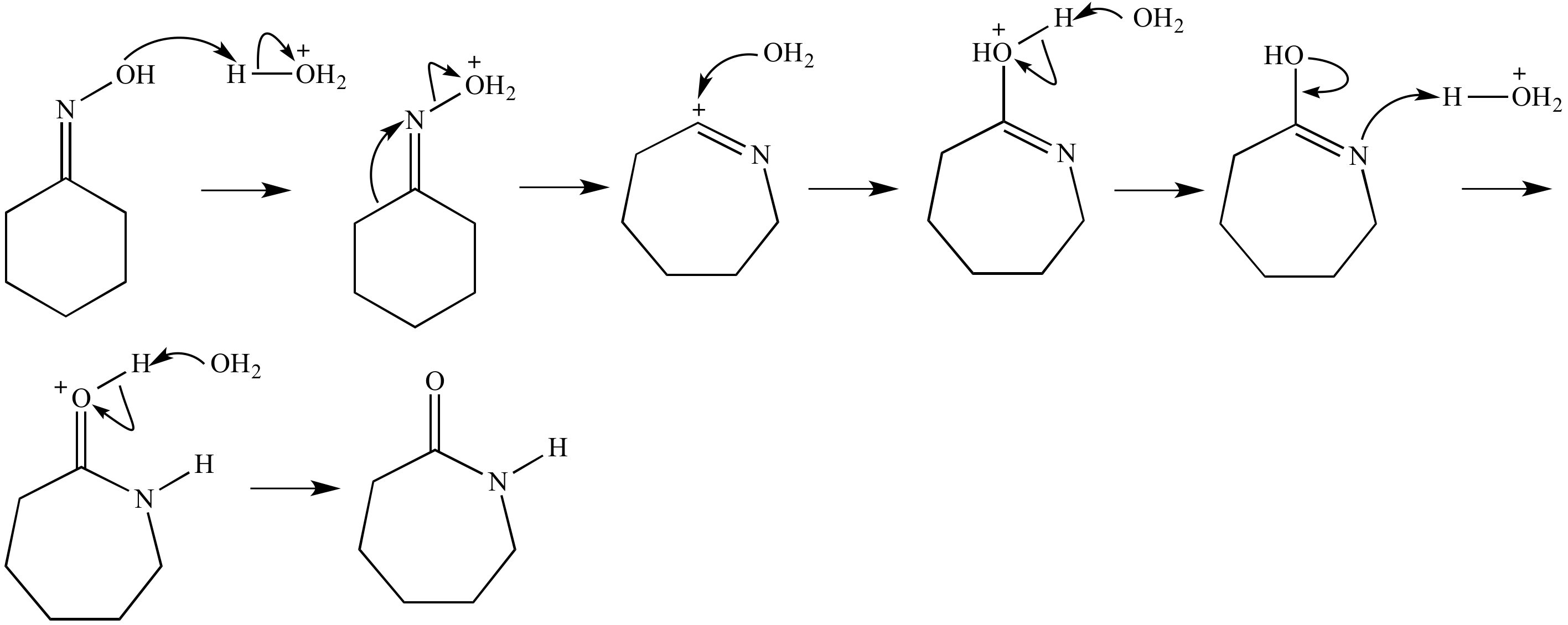
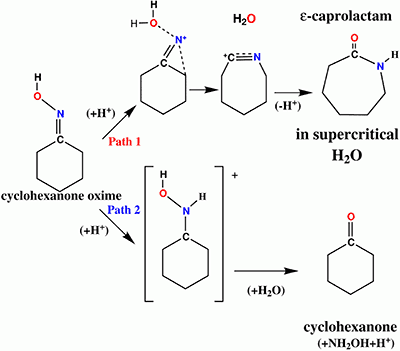
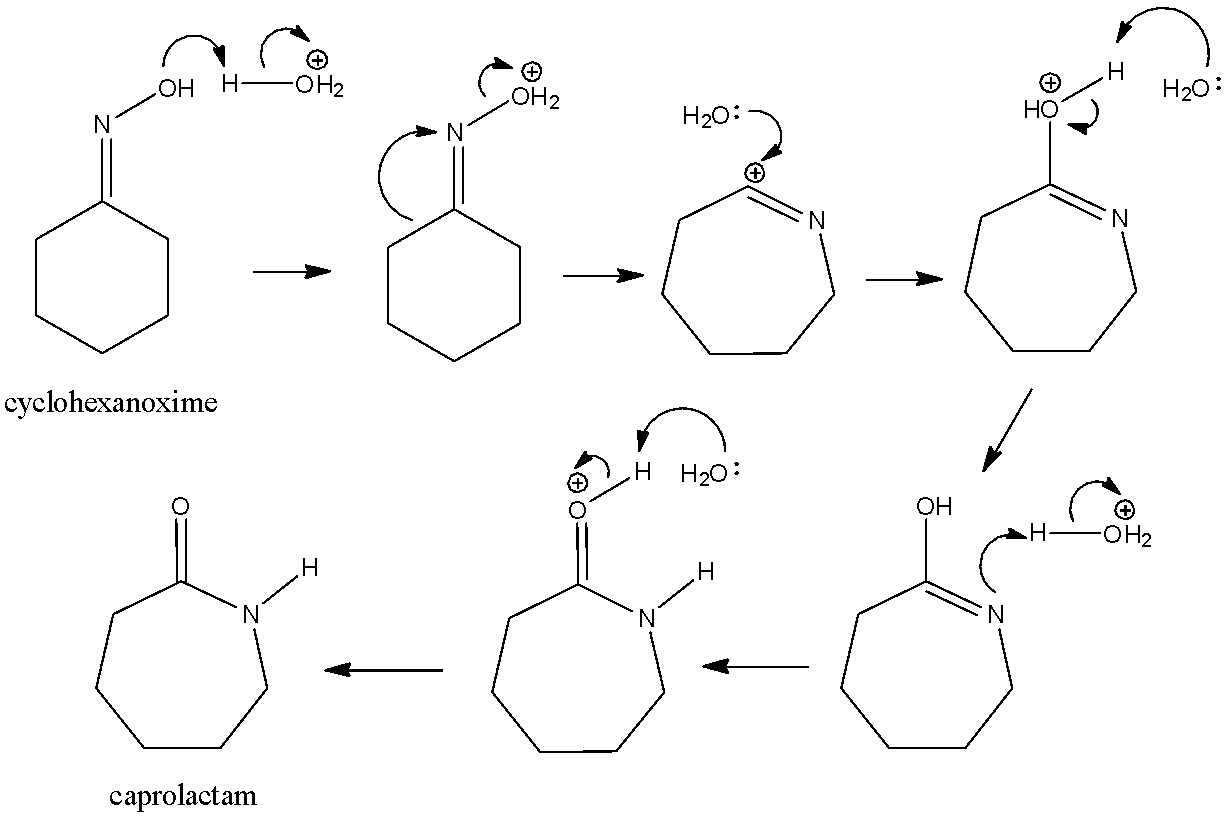
Beckmann rearrangement of cyclohexanone oxime in a microchemical system: The role of SO3 and product inhibition - Zhang - 2012 - AIChE Journal - Wiley Online Library

Application of an immobilized ionic liquid for the preparation of hydroxylamine via hydrolysis of cyclohexanone oxime - Wang - 2021 - Zeitschrift für anorganische und allgemeine Chemie - Wiley Online Library
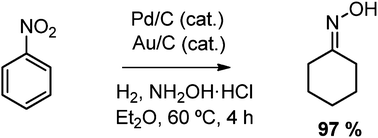
One pot synthesis of cyclohexanone oxime from nitrobenzene using a bifunctional catalyst - Chemical Communications (RSC Publishing)
![Beckmann Rearrangement of 15N-Cyclohexanone Oxime on Zeolites Silicalite-1, H-ZSM-5, and H-[B]ZSM-5 Studied by Solid-State NMR Spectroscopy | Journal of the American Chemical Society Beckmann Rearrangement of 15N-Cyclohexanone Oxime on Zeolites Silicalite-1, H-ZSM-5, and H-[B]ZSM-5 Studied by Solid-State NMR Spectroscopy | Journal of the American Chemical Society](https://pubs.acs.org/cms/10.1021/ja066392c/asset/images/large/ja066392cn00001.jpeg)
Beckmann Rearrangement of 15N-Cyclohexanone Oxime on Zeolites Silicalite-1, H-ZSM-5, and H-[B]ZSM-5 Studied by Solid-State NMR Spectroscopy | Journal of the American Chemical Society

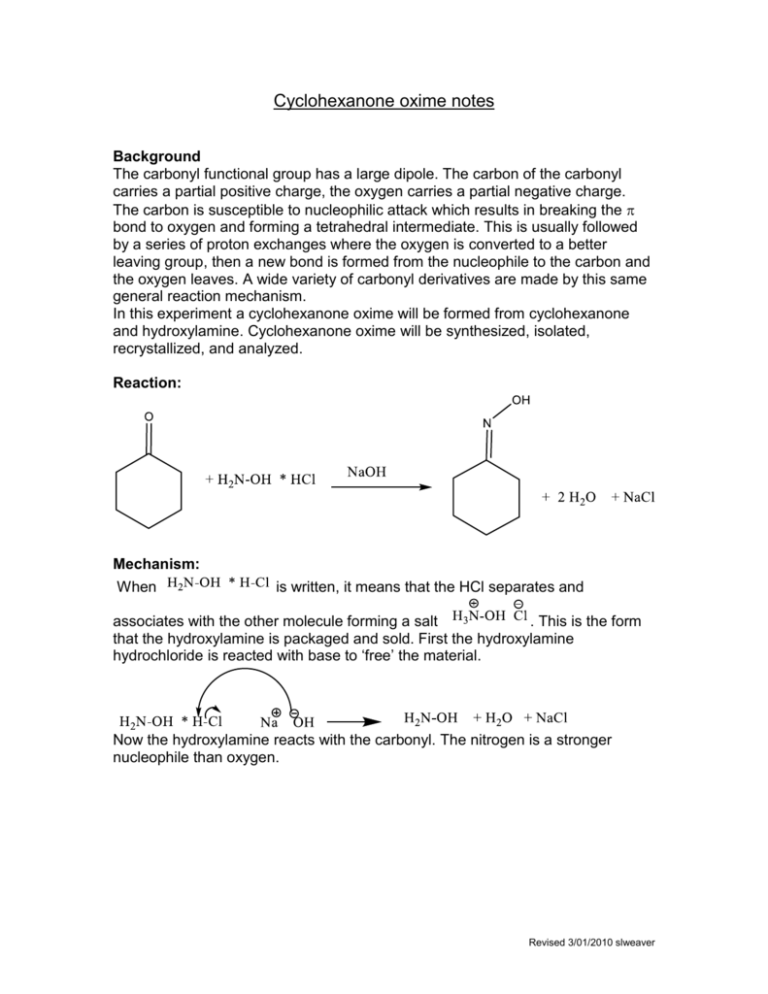
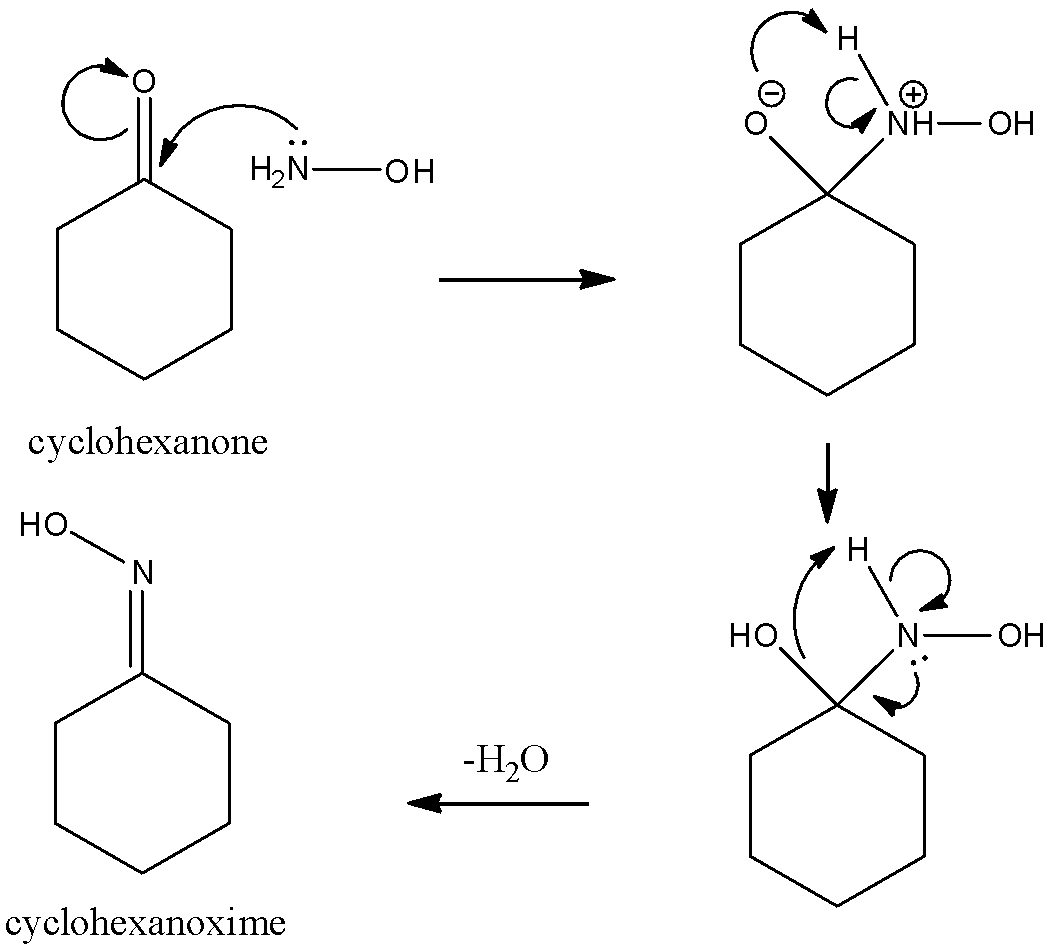
EXP Cyclohexanone Oxime and Its Rearrangement to -Caprolactam - Experiment 4 Cyclohexanone Oxime - Studocu

Beckmann rearrangement over phosphotungstic acid/SiMCM-41 cyclohexanone oxime to ε-caprolactam - ScienceDirect
![Provide a detalied mechanism for the following Rxn [{Image src='currentproblem7510002552455744196.jpg' alt='' caption=''}] 1. NH2OH \rightarrow 2. H3O^{+} | Homework.Study.com Provide a detalied mechanism for the following Rxn [{Image src='currentproblem7510002552455744196.jpg' alt='' caption=''}] 1. NH2OH \rightarrow 2. H3O^{+} | Homework.Study.com](https://homework.study.com/cimages/multimages/16/screen_shot_2019-11-10_at_7.56.28_pm5381447725266143897.png)
Provide a detalied mechanism for the following Rxn [{Image src='currentproblem7510002552455744196.jpg' alt='' caption=''}] 1. NH2OH \rightarrow 2. H3O^{+} | Homework.Study.com
![PDF] A DFT study on secondary reaction pathways in the acid-catalysed Beckmann rearrangement of cyclohexanone oxime in aprotic solvent☆ | Semantic Scholar PDF] A DFT study on secondary reaction pathways in the acid-catalysed Beckmann rearrangement of cyclohexanone oxime in aprotic solvent☆ | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/55d436b7cfa7a77d65878c0691e12cbd4fc73af2/2-Figure1-1.png)
PDF] A DFT study on secondary reaction pathways in the acid-catalysed Beckmann rearrangement of cyclohexanone oxime in aprotic solvent☆ | Semantic Scholar
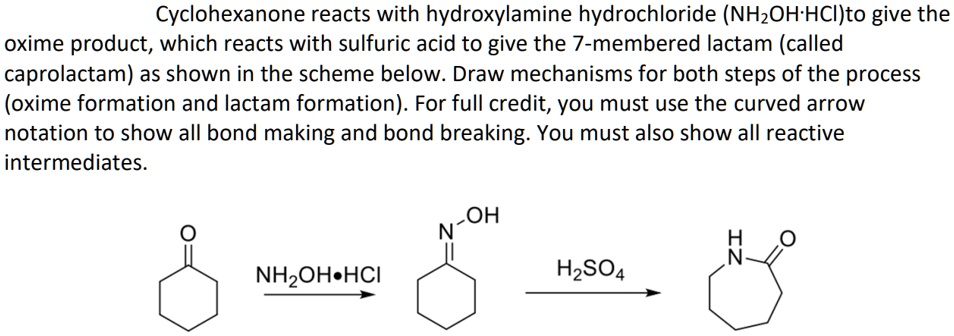
SOLVED: Cyclohexanone reacts with hydroxylamine hydrochloride (NHzOHHCIJto give the oxime product, which reacts with sulfuric acid to give the 7-membered lactam (called caprolactam) as shown in the scheme below. Draw mechanisms for

A new procedure to obtain ε-caprolactam catalyzed by a guanidinium salt - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C7NJ03443A